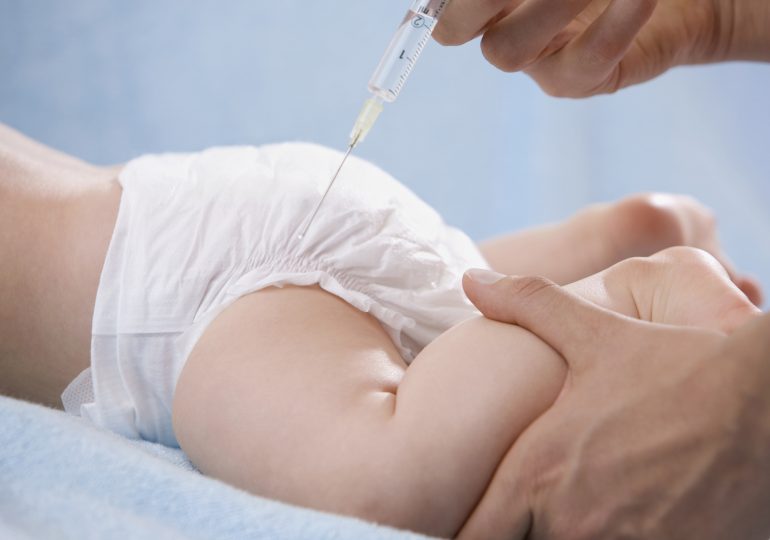
Doctors and parents celebrated the major advances that came in 2023 to treat respiratory syncytial virus (RSV), which sends up to 80,000 children under age five to the hospital each year in the U.S. This year, the U.S. Food and Drug Administration approved two critical ways to reduce the risk of RSV in young kids: a vaccine for pregnant mothers that can protect newborns, and a drug treatment for babies under one year.
[time-brightcove not-tgx=”true”]
In a study published in the New England Journal of Medicine, researchers report encouraging real-world data that show how effective the drug treatment, nirsevimab (brand name: Beyfortus), can be. The study, which was funded by the drug’s makers Sanofi and AstraZeneca, included more than 8,000 infants in France, Germany, and the U.K. who were one year old or younger and entering their first RSV season, which runs from fall to spring. Nivrsevimab is a monoclonal antibody that acts almost like a vaccine, training a child’s immune system to recognize RSV and defend against it. Half of the babies in the study were randomly assigned to receive nirsevimab, and half received no treatment. The drug was 83% effective in preventing hospitalization among those getting it, and 75% effective in reducing severe RSV. These results were similar regardless of the baby’s age, gestational age, or sex.
“From a scientific point of view, it’s fantastic that were able to prevent more than 80% of children who received the drug from going to the hospital,” says Dr. Saul Faust, pediatrician at University Hospital Southampton and co-leader of the study.
The findings support recommendations by the U.S. Centers for Disease Control and Prevention (CDC) that any baby under 8 months old receive a single injection of nirsevimab before their first RSV season if the mother has not already been vaccinated against RSV. But since the drug was approved in July, its makers, AstraZeneca and Sanofi, have not been able to keep up with the surge in demand. “The demand for Beyfortus has far surpassed any previous standard,” an AstraZeneca spokesperson said in response to questions about ongoing issues with supply.
In October, the CDC alerted doctors about ways to restrict supply of nirsevimab to babies at highest risk of RSV complications, such as younger and lower weight babies, and those born with underlying health conditions. The agency also recommended that some of these high-risk babies, including those born prematurely and who have heart and lung conditions, continue to receive an older, existing RSV treatment called palivizumab (brand name: Synagis). That can keep supplies of nirsevimab available for babies who don’t qualify for palivizumab. While paliviziumab is safe and effective, it requires monthly injections throughout RSV season, so doctors and parents were hoping to start taking advantage of nirsevimab’s single-injection.
For families with babies who aren’t at especially high risk of RSV complications, Dr. Rick Malley, a pediatrician at Harvard Medical School and Boston Children’s Hospital, says there are ways to protect infants even if they can’t get the shot. Especially during the holidays, Malley says parents can ask visitors to avoid exposing themselves to the baby if they have any symptoms of respiratory infections, such as a fever, cough, or runny nose. If visitors insist on getting close to the baby, touching the infant’s feet rather than face can also reduce the risk of passing on any infections.
The other reassuring news is that in general, RSV infections tend to peak earlier in the season, so it’s possible the highest risk is over for this year. Still, nirsevimab’s manufacturer anticipates that supplies will gradually continue to increase in coming months. “Approximately 230,000 additional doses will be made available in mid-January for this RSV season,” says the AstraZeneca spokesperson. “This follows the announcement in November that 77,000 additional doses were being made available.”
Leave a comment